Leading European diabetes experts from twenty academic research institutions and five pharmaceutical research organizations launched the DIRECT project in February, 2012. Project duration was seven years, with formal funding ending in July 2019.
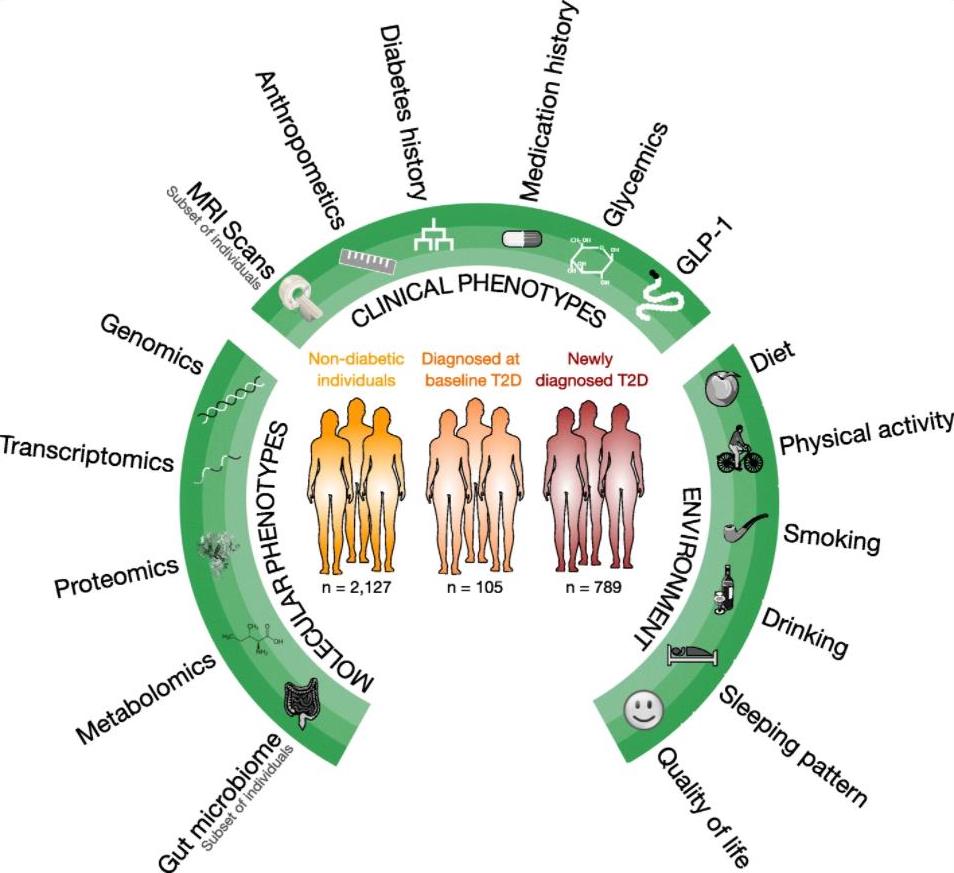
Image courtesy of Helle Krogh Pedersen, University of Copenhagen
The overarching aims of the DIRECT consortium are to identify potential biomarkers that will help identify patients whose disease may progress more rapidly or patients who have a different response to diabetes treatments. We thereby hope to develop a ‘personalised’ or ’stratified’ medicines approach to treatment of type 2 diabetes’ (T2D) using either existing or novel therapies.
The DIRECT consortium addressed two fundamental areas of heterogeneity of diabetes:
- The rate at which people´s glycaemia deteriorates either from pre-diabetes to diabetes, or through diabetes with increasing treatment requirements
- The extent to which certain therapeutic interventions (either drug or surgical) results in improvement in glycaemia.
In accordance with the IMI research agenda the DIRECT consortium offers considerable potential to achieve major progress towards a personalized medicines approach for the treatment of type 2 diabetes.
The close cooperation of academic teams and pharmaceutical companies in the DIRECT project provided a unique platform of expertise in a Public-Private-Partnership between the European Commission (EC) and the European Federation of Pharmaceutical Industries and Associations (EFPIA) to improve industrial competitiveness and Public Health in Europe.
For more information please see the websites of IMI, EFPIA and the European Commission.
The IMI Diabetes Platform
Collaborative approaches to overcome key bottlenecks on the way to novel therapies
A state-of-the-art flyer titled “The IMI Diabetes Platform” was released in 2012 together with two other IMI diabetes consortia, SUMMIT and IMIDIA.
The flyer was designed to give both visibility to each individual consortium and at the same time a good understanding of those aspects of IMI diabetes research that together constitute one of the most holistic discovery approaches in diabetes to date.
The IMI diabetes flyer highlights unique selling points and covers topics for those with a special interest in IMI diabetes projects, e.g. academia, pharma, the EC and other stakeholders like country organizations or members state groups, clinicians, patients, economists, national health authorities as well as patient organizations.
The flyer was first presented to a broad scientific audience at the 2012 EASD annual meeting in Berlin via the EASD information booth and DIRECT EFPIA participant stands at the exhibition.
About IMI
The Innovative Medicines Initiative (IMI)
The DIRECT project is supported by the Innovative Medicines Initiative (IMI), a large-scale public-private partnership between the European Union and the European Federation of Pharmaceutical Industries and Associations (EFPIA) which as the names indicates is an association of pharmaceutical companies in Europe.
IMI aims to boost biopharmaceutical innovation in Europe and to speed up the development of better and safer medicines for patients. Through its unique and innovative funding scheme, IMI supports research projects in the areas of safety and efficacy, knowledge management and education and training.
In addition to the large biopharmaceutical companies, the participating research consortia include small- and medium-sized enterprises, patient organisations, academia and other research organisations, hospitals and public authorities.



